
The same concept applies to the other columns of the periodic table. The organization of electrons in atoms explains not only the shape of the periodic table, but also the fact that elements in the same column of the periodic table have similar chemistry. Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistry- and they do. They all have a similar electron configuration in their valence shells: a single s electron. Their electron configurations (abbreviated for the larger atoms) are as follows, with the valence shell electron configuration highlighted: Electrons, electron configurations, and the valence shell electron configuration highlighted. For example, take the elements in the first column of the periodic table: H, Li, Na, K, Rb, and Cs. Match the atoms on the periodic table with the statements regarding the ground state electron configuration Afilled set of sorbitals and a partially filled set of porbitals BA filled set of dorbitals cA half-filled set of dorbitals DA filled set of and porbitals in the valence shell E One electron.
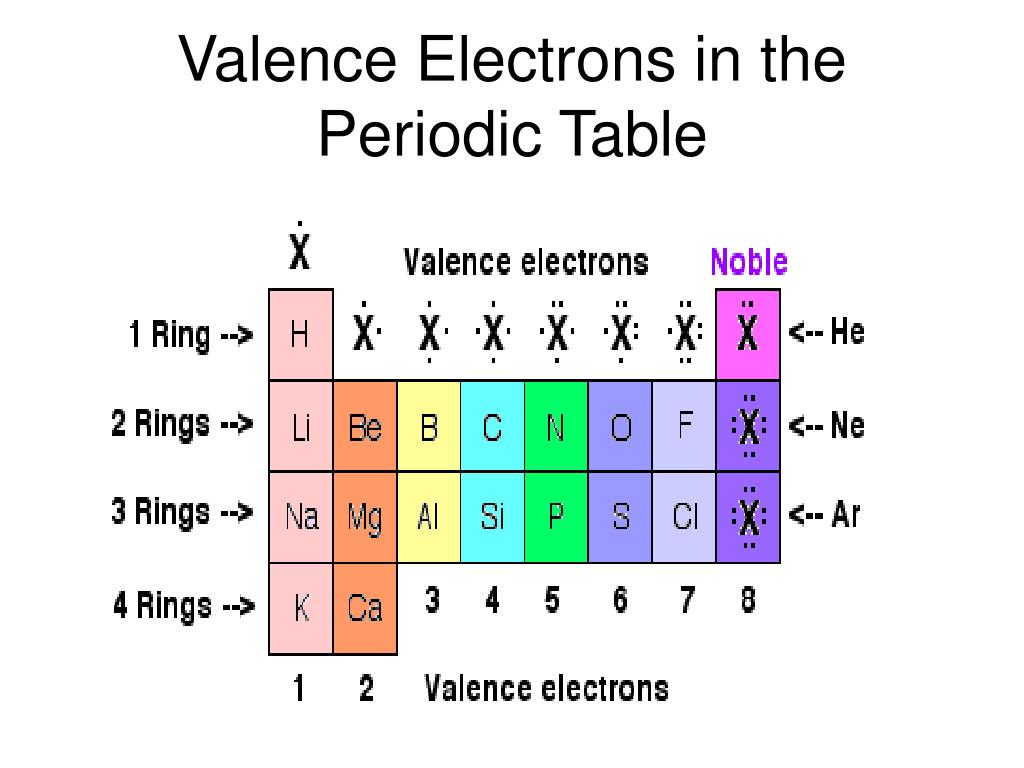
If we look at just the valence shell’s electron configuration, we find that in each column, the valence shell’s electron configuration is the same. (The inner electrons are called core electrons.) The valence electrons largely control the chemistry of an atom. The electrons in the highest-numbered shell, plus any electrons in the last unfilled subshell, are called valence electrons the highest-numbered shell is called the valence shell. The periodic table is separated into blocks depending on which subshell is being filled for the atoms that belong in that section.
